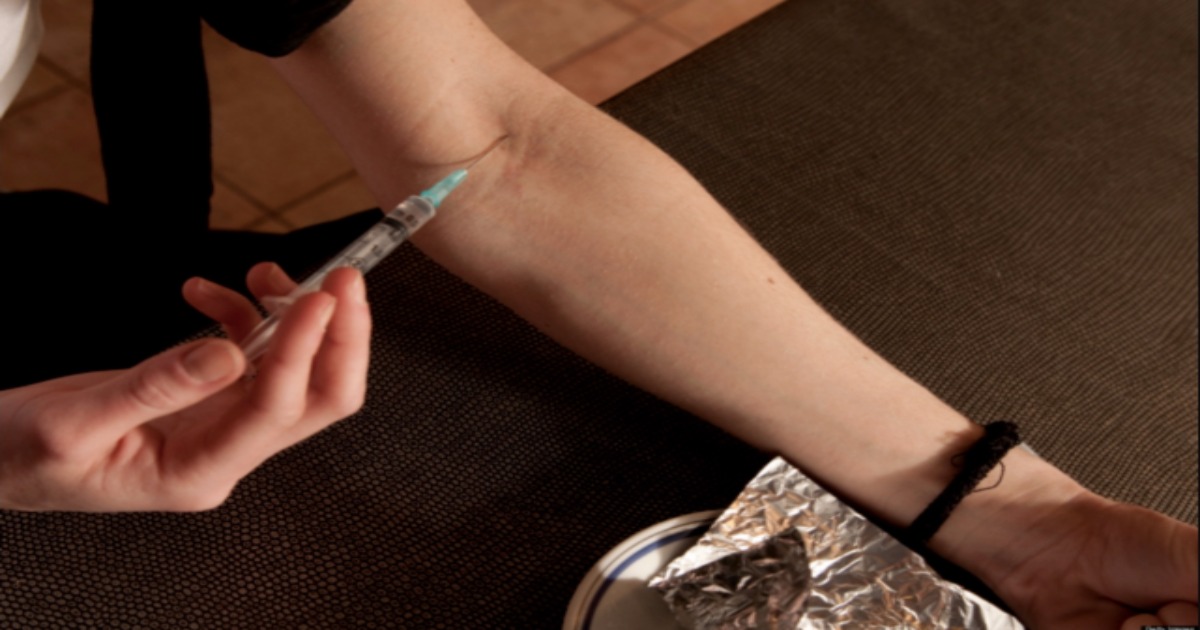
Drug linked to HIV Outbreak will Likely Stay on the Market
The risk associated with abusing Opana ER is greater than its benefits as a painkiller, according to a panel of experts at a joint FDA committee meeting. A press release from Endo, the manufacturer of Opana detailed the ruling: “The U.S. Food and Drug Administration’s (FDA) Drug Safety Risk Management and Anesthetic and Analgesic Drug Products Advisory Committees voted 18 to eight, with one abstention, that the benefits of reformulated OPANA® ER (oxymorphone hydrochloride extended release) no longer outweigh its risks. While several of the Advisory Committee members acknowledged the role of OPANA® ER in clinical practice, others believed its...
Read More